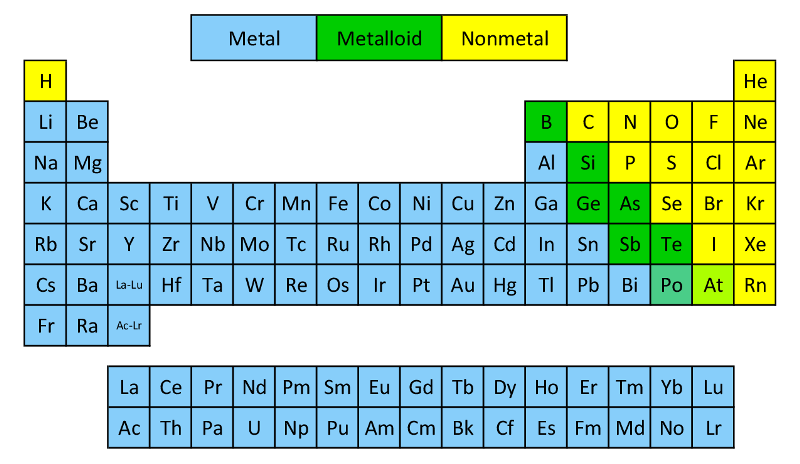
In the standard layout of the periodic table, metalloids occur along the diagonal line through the p block from boron to astatine. Phosphorus, tin, selenium and bismuth also have allotropes which display borderline behavior. For example, for the element carbon, its diamond allotrope is clearly non-metallic, however the graphite allotrope displays limited electric conductivity more characteristic of a metalloid. Some allotropes of elements exhibit more pronounced metal, metalloid or non-metal behavior than others. The following elements are generally considered metalloids: Semiconductor refers to the physical properties of materials (including alloys, compounds) and there is only partial overlap between the two. Metalloid refers to the properties of certain elements in relation to the periodic table. The concepts of metalloid and semiconductor should not be confused. metalloids often behave as semiconductors (B,Si,Ge) to semimetals (eg.metalloids often form amphoteric oxides.There is no rigorous definition of the term, however the following properties are usually considered characteristic of metalloids: (In Greek metallon = metal and eidos = sort) On the basis of their general physical and chemical properties, nearly every element in the periodic table can be termed either a metal or a nonmetal - however a few elements with intermediate properties are referred to as metalloids. Metalloid is a term used in chemistry when classifying the chemical elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
